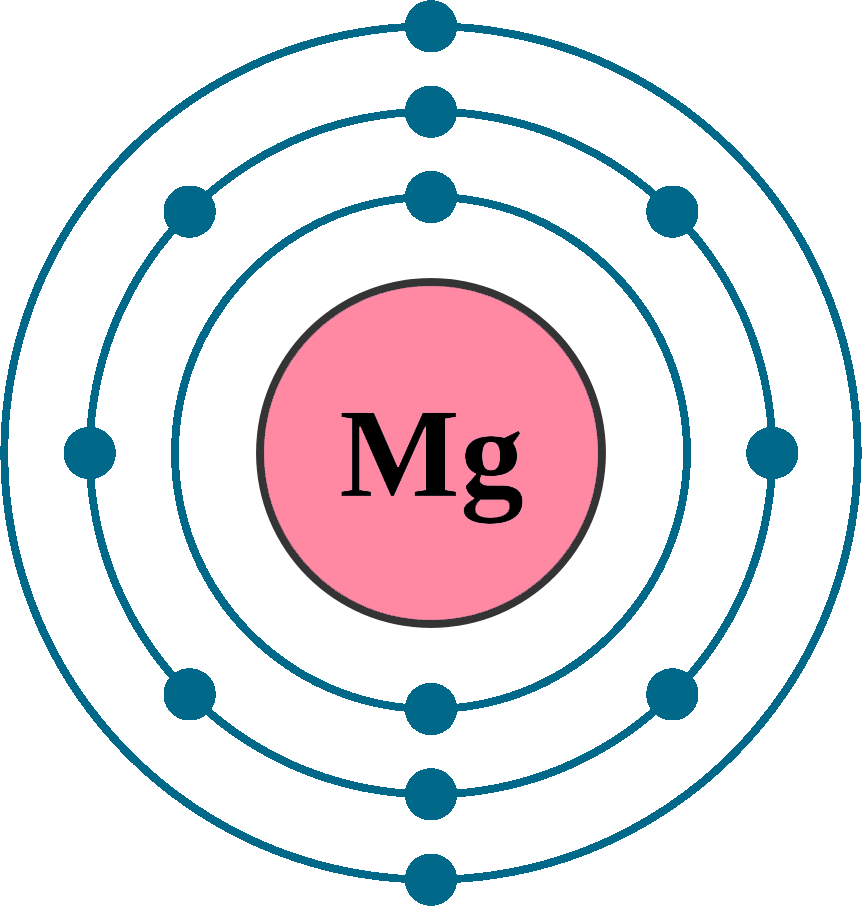
Magnesium Electrons Charge . The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. (b) since n 3− is an anion, its name. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. To get a +2 charge, the ion had to lose two electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. The symbol for the ion is mg 2+ , and it is called a. The symbol for the ion is mg 2+ , and it is. A magnesium ion therefore has 10 electrons. It describes how electrons are distributed. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals.
from mungfali.com
The symbol for the ion is mg 2+ , and it is. The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. A magnesium ion therefore has 10 electrons. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. (b) since n 3− is an anion, its name. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is mg 2+ , and it is called a. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. To get a +2 charge, the ion had to lose two electrons.
Magnesium Aufbau Diagram
Magnesium Electrons Charge To get a +2 charge, the ion had to lose two electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is mg 2+ , and it is called a. A magnesium ion therefore has 10 electrons. The symbol for the ion is mg 2+ , and it is. To get a +2 charge, the ion had to lose two electrons. It describes how electrons are distributed. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. (b) since n 3− is an anion, its name. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+.
From utedzz.blogspot.com
Periodic Table Magnesium Electron Configuration Periodic Table Timeline Magnesium Electrons Charge Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. To get a +2 charge, the ion had to lose two electrons. The symbol for the ion is mg 2+ , and it is. The. Magnesium Electrons Charge.
From www.youtube.com
Mg Orbital Diagram How to Write the Atomic Orbital Diagram for Magnesium Electrons Charge The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. The symbol for the ion is mg 2+ , and it is called a. The symbol for the ion is mg 2+ , and it is. (b) since n 3− is an anion, its name. It describes. Magnesium Electrons Charge.
From www.thesciencehive.co.uk
Atomic Structure and Electron Configuration (AQA) — the science hive Magnesium Electrons Charge The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. It describes how electrons are distributed. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is mg 2+ , and it is. Magnesium Electrons Charge.
From www.alamy.com
3d render of atom structure of magnesium isolated over white background Magnesium Electrons Charge (b) since n 3− is an anion, its name. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. It describes how electrons are distributed. The symbol for the ion is mg 2+ , and it is called a. The symbol for the ion is mg 2+ , and it is. The first person. Magnesium Electrons Charge.
From www.youtube.com
Draw the Lewis Structure of MgBr2 (magnesium bromide) YouTube Magnesium Electrons Charge Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. (b) since n 3− is an anion, its name. The symbol for the ion is mg 2+ , and it is. A magnesium ion therefore has 10 electrons. To get a +2 charge, the ion had to lose two electrons. Thus,. Magnesium Electrons Charge.
From www.sciencephoto.com
Magnesium, atomic structure Stock Image C018/3693 Science Photo Library Magnesium Electrons Charge It describes how electrons are distributed. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. The symbol for the ion is mg 2+ , and it is called a. A magnesium ion therefore has 10 electrons. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. To. Magnesium Electrons Charge.
From www.dreamstime.com
Diagram Representation Of The Element Magnesium Stock Vector Image Magnesium Electrons Charge It describes how electrons are distributed. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. The symbol for the ion is mg 2+ , and it is. Thus, a magnesium atom will form a cation with two. Magnesium Electrons Charge.
From www.myxxgirl.com
Orbital Diagram Of Magnesium Wiring Diagrams Manual My XXX Hot Girl Magnesium Electrons Charge These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. A magnesium ion therefore has 10 electrons. It describes how electrons are distributed. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. To get a +2 charge, the ion had to lose two electrons. The first person. Magnesium Electrons Charge.
From userlibschwartz.z19.web.core.windows.net
Magnesium Electric Dot Diagram Magnesium Electrons Charge A magnesium ion therefore has 10 electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. It describes how electrons are distributed. To get a +2 charge,. Magnesium Electrons Charge.
From sciencenotes.org
Magnesium Atom Science Notes and Projects Magnesium Electrons Charge To get a +2 charge, the ion had to lose two electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. Thus, a magnesium atom will form a cation with two fewer electrons than. Magnesium Electrons Charge.
From www.toppr.com
How is magnesium chloride formed by the transfer of electrons? Why does Magnesium Electrons Charge A magnesium ion therefore has 10 electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is mg 2+ , and it is called a. To get a +2 charge, the ion had to lose two electrons. The first person to propose that magnesium was. Magnesium Electrons Charge.
From valenceelectrons.com
How to Find the Valence Electrons for Copper (Cu)? Magnesium Electrons Charge The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. (b) since n 3− is an anion, its name. The symbol for the ion is mg 2+ , and it is.. Magnesium Electrons Charge.
From valenceelectrons.com
How Many Protons,Neutrons and Electrons Does Magnesium Have? Magnesium Electrons Charge To get a +2 charge, the ion had to lose two electrons. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. It describes how electrons are distributed. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the. Magnesium Electrons Charge.
From periodiske-system.dk
Magnesium Magnesium Electrons Charge These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. The symbol for the ion is mg 2+ , and it is called a. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. (b) since n 3− is an anion, its name. Thus, a magnesium atom will. Magnesium Electrons Charge.
From slidetodoc.com
Ionic Bonding Elements are the simplest substances There Magnesium Electrons Charge It describes how electrons are distributed. The symbol for the ion is mg 2+ , and it is called a. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. (b) since n 3− is an anion, its name. A magnesium ion therefore has 10 electrons. The first person to propose. Magnesium Electrons Charge.
From www.animalia-life.club
Magnesium Electron Configuration Magnesium Electrons Charge (b) since n 3− is an anion, its name. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate, magnesium chloride, and magnesium citrate. To get a +2 charge, the ion had to lose two electrons. Thus, a magnesium atom will form a. Magnesium Electrons Charge.
From utedzz.blogspot.com
Periodic Table Magnesium Electron Configuration Periodic Table Timeline Magnesium Electrons Charge A magnesium ion therefore has 10 electrons. It describes how electrons are distributed. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The electron configuration of magnesium refers to the arrangement of electrons in the magnesium atom’s orbitals. The symbol for the ion is mg 2+ , and it is.. Magnesium Electrons Charge.
From ar.inspiredpencil.com
Magnesium Chemical Symbol Magnesium Electrons Charge A magnesium ion therefore has 10 electrons. The first person to propose that magnesium was an element was joseph black of edinburgh in 1755, and an impure form of metallic. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. These forms of magnesium can range from magnesium hydroxide, magnesium sulfate,. Magnesium Electrons Charge.